Leading care forward by advocating for our customers and their patients
HCHB Responds to CMS on the 2024 Proposed Rule on Behalf of the Home-Based Care Industry
1st CHAP Dual Verified EHR

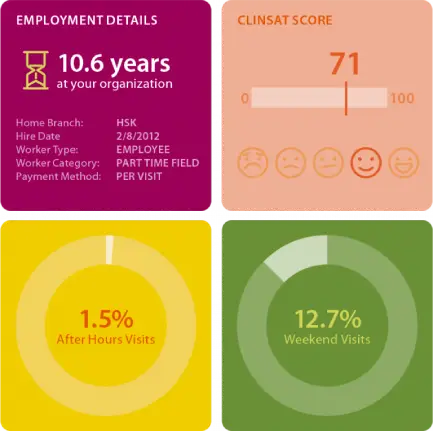
HCHB Analytics provides powerful insight into clinician satisfaction
HCHB Clinician Satisfaction Dashboard provides a data-driven view of satisfaction, gathers insights, and identifies at-risk of turnover.

Quickly capture accurate, detailed documentation at every visit
HCHB offers powerful solutions for home-based care agencies of all sizes

Put complex scheduling on autopilot
Get advanced interoperability across the entire healthcare community with HCHB Connect
Home Care Software Designed By Nurses, For Nurses
Get the software and tools you need to boost patient care, satisfaction and outcomes. Watch the video overview below to see how we can help you!
We’re the perfect fit for home health or hospice agencies of any size

The HCHB platform puts smart solutions right at your fingertips
Our powerful software streamlines every aspect of your business for maximum productivity and long-term success.
HCHB Services help you maximize resources and boost productivity
Make the most of your software investment with added services that can ease the workload.
Use HCHB Intelligence to leverage critical data and insight
Use critical data to keep your agency running smarter and help make informed business decisions.
HCHB Connect allows seamless sharing of complex data
Stay connected with interoperability tools to enhance patient care across every touch point.
Get Started Today with Homecare Homebase
NEWS HIGHLIGHTS

CMS Releases 2025 Hospice Proposed Rule
March 29, 2024

HCHB Customers in the News: April 2023
April 17, 2023

HCHB Customers in the News: March 2023
March 2, 2023

HCHB Customers in the News: February 2023
February 6, 2023
Blog HIGHLIGHTS

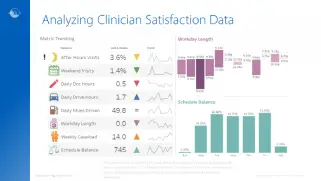
Using HCHB Analytics to Improve Clinician Satisfaction
December 7, 2022
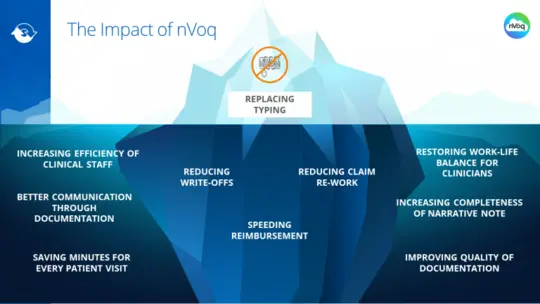
The Impact of Speech Recognition Solutions on Post Acute Documentation
November 10, 2022

Streamline the Home Health EVV Process with EVVLink
November 10, 2022